Cardiology Case #21
Primary Author: Dr Alastair Robertson; Co-Authors: Dr Hywel James and David Law
Background:
A 64yo man presents to a regional ED with generalised fatigue over several weeks, then worsening breathlessness over the last 4 days. He describes orthopnoea and some leg swelling. He has not had any fevers or infectious symptoms. He discloses very little previous medical history.
On examination he has bibasal creps & mild pitting oedema bilaterally.
His observations are as follows:
RR 20 Sats 91% on air HR 130 BP 133/86 temp 36.7
What do you think of his Chest XR and ECG (below)
-
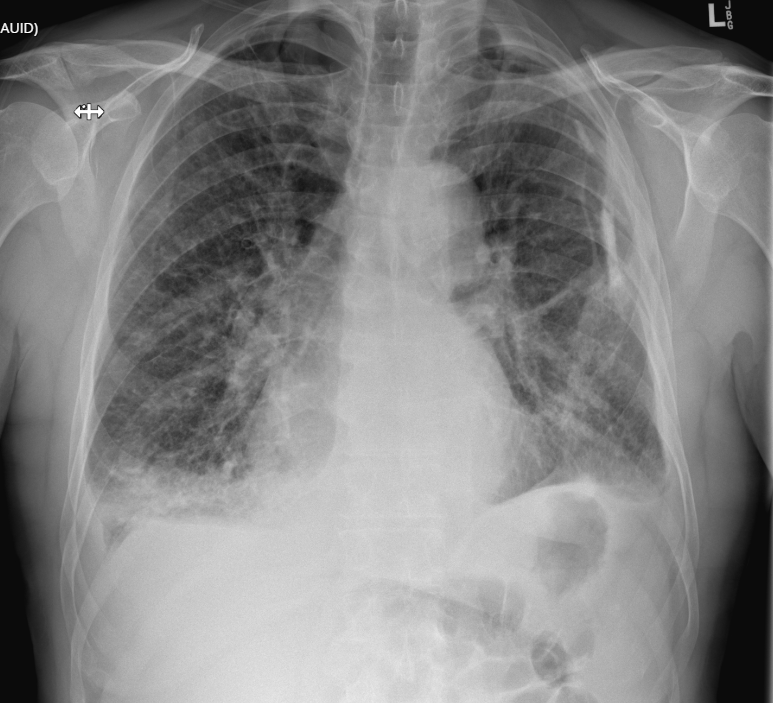
CXR shows:
bilateral pleural effusions, with bilateral airspace opacification suggesting pulmonary congestion.
Left sided pleural calcification is noted.
Possible right upper lobe consolidation.
Overall in keeping with congestive cardiac failure.
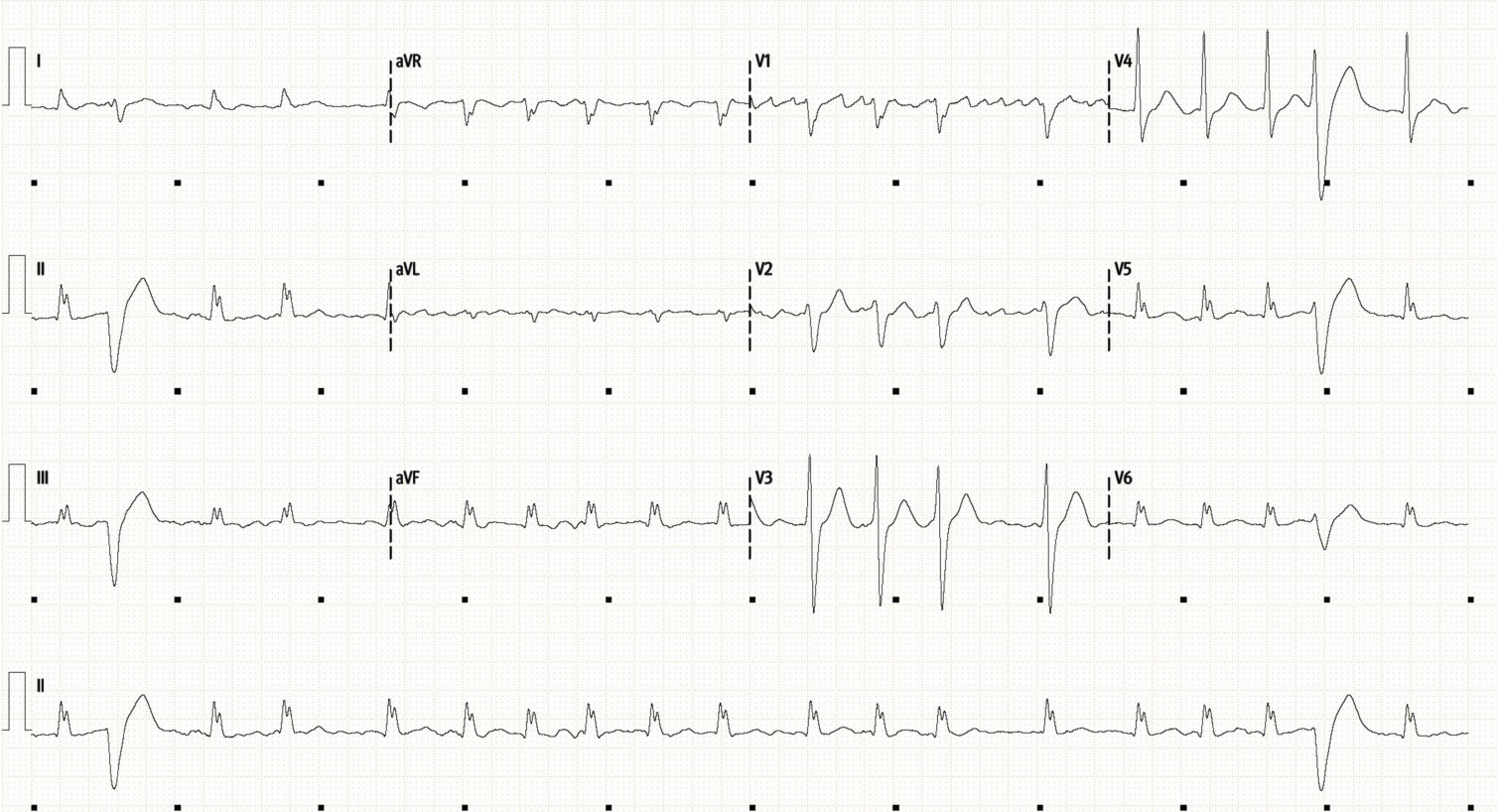
ECG shows:
Atrial Fibrillation with an irregular tacyhcardia of around 115/min with some PVCs. Course fibrillation waves can be seen in V1 which can often appear quite organised in the right-sided leads in AF.
The QRS is borderline broad, at 120ms with a left bundle morphology, just about meeting criteria for a LBBB.
Cardiac POCUS:
This is an otherwise previously well patient presenting with features of cardiac failure and a tachyarrythmia. We have seen several cases similar to this previously where POCUS has shown significant structural cardiac disease so it is important to assess for this, as well as assessing LV function to help guide our management.
Below are the Parastenal Long Axis (PLAX), a zoomed view of the mitral valve (PLAX), and a short-axis at the mid-cavity (SAX).
Can you interpret the PLAX looking at all the review areas?
What do you think of the LV function?
What do you think of the mitral valve?
Basic POCUS: global assessment
PLAX shows:
no pericardial effusion or obvious pleural effusion
The LV looks moderately dilated, and formal echo confirmed moderate LV dilation (LVDd 6.3cm). The patient is in AF so there is beat-to-beat variability but the systolic function is clearly impaired. This is moderate-to-severe global impairment, with EF estimated at 30% (severe is EF <30%, moderate is EF 30-40%). This improved slightly as his rate was controlled, to become moderate impairment.
Looking at RV:Ao:LA ratio, the Right ventricle looks normal, there is moderate dilation of the aortic root (measured at 4.9cm, see Case 20 for a review of Aorta). The left atrium is severely dilated, we can see it is almost the same size as the left ventricle. This suggests an element of chronicity to the pathology.
The aortic valve looks normal, leaflets are opening well which excludes critical aortic stenosis.
The mitral valve is very abnormal, and can be seen in further detail on the zoom view (middle clip). There is gross thickening and fibrosis of both leaflets, with dense echo bright lesions attached to both the mitral valve leaflets.
SAX view, confirms the moderate-to-severe global LV impairment with no obvious regional wall motion abnormalities.
So in summary, this patient has a very abnormal mitral valve with dense mobile lesions on both valve leaflets which are concerning for vegetations secondary to infective endocarditis. We would suspect there may be significant mitral regurgitation associated with these lesions.
There is severe left atrial dilation suggesting that this process has been progressing for some time. He is in atrial fibrillation, with moderate to severe LV impairment, so may well have developed AF due to worsening mitral regurgitation, and is not tolerating it very well, hence his decompensation and presentation to ED.
Case Progress:
Having identified a clearly abnormal mitral valve with possible vegetations further examination, and a deeper review of previous notes was conducted.
The patient did not have any stigmata, either immunological or vascular phenomenon suggestive of IE.
Previous notes documented a presentation 10 years prior with suspected endocarditis but an incomplete course of IV antibiotics.
This raised suspicion for either active, or previous endocarditis.
Steps beyond Basic POCUS: Colour Doppler
The 2D imaging shows the dense vegetations on the mitral valve, and a severely dilated left atrium. We suspect there to be significant (probably severe) mitral regurgitation. To image this, use colour doppler to assess the regurgitant jet.
Ensure the colour box is placed over the whole of the left atrium to pick up ‘eccentric jets’ suggestive of Severe MR which wrap around the wall of the atrium.
Top - PLAX
Middle and bottom - A4C
In the top picture you can see a broad regurgitant jet. The colour doppler shows a complex array of regurgitation because the large vegetations cause multiple irregular orifices through which blood can regurgitate through.
You can see a narrow jet being directed inferiorly towards the left atrial wall which is an eccentric jet which likely wraps around the wall. These are typical of severe mitral regurgitation (see Case 17 for a review of eccentric MR jets).
The middle clip is colour doppler from the A4C view - here you can see a convergenge zone just above the mitral valve in the LV, and then an eccentric jet tracking towards the intraatrial septum (to the left of the screen) signifying severe MR.
The bottom clip is a 2D image of the same A4C view. Here you can see the dense vegetations on the mitral valve. Also look closely at the valve leaflets when they close. You can see the gap (called a coaptation defect) which allows the blood to flow (in an ‘eccentric direction’) towards the intraatrial septum. The left atrium is severely dilated (similar size to the LV) suggesting an element of chronicity to this presentation.
Intermediate POCUS:
Other key points to address when assessing patients with suspected IE
Severe MR often causes significant pulmonary hypertension. Ensure to assess pulmonary pressures by measuring the RVSP and IVC assessment.
Assessment of LV function is important - in severe MR the LV will usually be hyperdynamic. If the LV function is poor then this may represent a failing LV, particularly in the setting of low blood pressure or other features of shock.
Assessing other valves: if there is mitral valve involvement look for aortic valve endocarditis as well. Assess for aortic regurgitation and image the aortic valve to look for thickening or vegetations. It is also important to review the aortic root for aortic root abscess or aortitis. Below shows this patient had a normal appearance of the aortic valve, with only mild aortic regurgitation (narrow central jet).
Assessment of right-sided valves
It is also important to assess the right-sided valves. Particularly in IVDUs where tricuspid or pulmonary valve IE is more common.
The tricuspid valve is visualised from a parasternal window by tilting inferiorly from the PLAX, as well as from the apical 4-chamber view. Assess is as you would the mitral, looking for vegetations or severe regurgitation.
The Pulmonary valve can be difficult to visualise. It is best seen from a parasternal window as is it superior. From the PLAX view, tilt superiorly to visualise the valve. It is best seen however in the PSAX view of the aortic valve. The pulmonary valve and RVOT will be situated above and to the right of the aortic valve.
The video below shows a SAX view with the aortic valve in the centre. The pulmonary valve is situated top right of screen, at about 1 o’clock. Note the trivial pulmonary regurgitation. Image the valve in 2D to assess for vegetations and look for significant regurgitation using colour.
Case Progress:
The patient was diagnosed with Severe mitral regurgitation, secondary to healed vegetations on the mitral valve from previous infective endocarditis.
TOE was performed, more clearly showing the valve, and the MR (see clips below).
The patient was initially managed with diuresis to optimise fluid status, started on anticoagulation due to atrial fibrillation/flutter, and was referred for inpatient workup for a mitral valve replacement.
TOE:
Trans-oesophageal echo is important in the assessment of suspected infective endocarditis. Whilst TTE has high specificity, its sensitivity is poor and may miss small vegetations or perivalvular complications, and is less useful for detailed assessment of both native and prosthetic valves.
TOE also gives better assessment of valvular pathology such as regurgitation to assess the need for valve replacement.
See the TOE images (above) of the mitral valve. Given the TOE probe is in the oesophagus behind the heart, the chamber orientation has changed. The LV is at the bottom and the LA at the top of the screen. Dense, echobright lesions can be clearly seen on both valve leaflets. Also note the acoustic shadowing behind the vegetations due to calcium buildup.
The bottom clip shows the complex severe mitral regurgitation.
Imaging “Major” ISCVID modified Duke criteria (see below) which can be satisfied by echo are the presence of vegetations, valve perforation, abscess, or significant new regurgitation. The clinical criteria definitions are as follows:
“Echo and/or Cardiac CT showing vegetation, valvular/leaflet perforation or aneurysm, abscess, pseudoaneurysm, or intracardiac fistula.”
“Significant new valvular regurgitation as compared with previous imaging.”
FOCUS on: Infective Endocarditis & Echo
Infective endocarditis is a complex disease so we will aim to cover the key points for the ED physician.
Common Organisms
Staph aureus - often skin flora, IVDUs, indwelling devices
Strep spp. - S. viridians often associated with oral/dental sources. S. bovis is strongly associated with colorectal cancer.
Coag negative Staph (S. epidermidis) often infects prosthetic valves or devices.
Enterococcus spp. - often from GI source, common in elderly patients after surgical procedures or in setting of GI malignancy.
HACEK group - (Haemophilus, Aggregatibacter, Cardiobacterium, Eikenella, Kingella), rarer gram negative bacilli that grow slowly and are difficult to culture.
Coxiella - consider in those with Q fever risk factors, often a more indolent clinical course, blood cultures often negative, consider serology screening.
Fungal - consider in immunocompromised patients, IVDUs, ICU patients.
High-risk Populations
Consider IE in any patient with unexplained fevers and/or a new murmur, but especially so in the following patient groups:
Previous IE - one of the highest predictors of reccurence
Prosthetic valves, previous TAVI, implantable devices (e.g. PPM)
Congential heart disease, especially if bicuspid aortic valve, or previous surgical repair.
IVDUs (typically Staph aureus, most commonly tricuspid valve)
Those on haemodialysis or with long-term indwelling access devices (e.g. portacaths, Hickman lines).
Those with poor dentition.
Elderly patients with degenerative valve disease
Immunocompromised.
Diagnosis has classically been with the Duke criteria, which use a combination of microbiological, imaging, and clinical criteria. These have been recently updated (to the 2023 ISCVID Criteria) - see here for more information on the current definition.
It is important to collect several blood cultures from different sites prior to initiation of antibiotics if possible. Ideally at least 3 sets of blood cultures should be drawn from peripheral sites (avoid using indwelling lines in the first instance).
Other important blood tests include inflammatory markers (WCC/CRP/ESR), haemolysis screen (blood film, LDH, urine microscopy), renal function (assess for renal infarction or glomerulonephritis), Q-fever serology, LFTs (septic hepatic emboli), troponin (demand ischaemia or septic MI), and rheumatological screen.
FOCUS on: IE
What to look for on echo:
There are four key features to look for on echo regarding infective endocarditis. In ED the primary goals are to have a low clinical threshold to suspect the disease, and to identify any obvious vegetations and/or significant regurgitation.
Formal TTE will assess for:
Vegetations: Typically oscillating, irregular masses attached to valves or endocardial surfaces. Large size (>10 mm) and very mobile vegetations are key predictors of embolic risk.
Abscess/Pseudoaneurysm: Thickened, echo-lucent areas within the perivalvular tissue, most commonly involving the aortic root. These can develop rapidly, and can also cause paravalvular regurgitation.
Leaflet Perforation: Often seen as a "jet" of regurgitation originating from the body of the valve leaflet rather than the coaptation line.
New Valvular Dehiscence: Specifically relevant in prosthetic valves, often manifesting as a "rocking" motion of the valve prosthesis.
Initial management of IE in the ED:
Most patients with IE will be relatively stable
ED management in general focusses on having a high clinical suspicion for the diagnosis (it is difficult to make a ‘definite’ diagnosis of IE in ED via the Duke criteria without microbiology, however it is possible with an echo positive for IE) and ensuring that multiple blood cultures have been collected and the correct investigations ordered.
For the unstable patient: you need to work out why they are unstable:
Sepsis: broad spectrum antibiotics:
Gentamicin + Flucloxacillin + Benzylpenicillin first line empiric therapy
Gentamicin + Flucloxacillin + Vancomycin (if septic/unstable, if prosthetic valve/indwelling devices, or if MRSA suspected)
Gentamicin + Vancomycin + Cefazolin (if allergic to penicillin)
Acute severe regurgitation: managed as described previously on here:
respiratory support as needed - HFNP or NIV
allow tachycardia 90-120 and expect a hyperdynamic LV
Target the lowest acceptable BP to minimise afterload
Avoid arrhythmia, consider treating AF aggressively as patients will often not tolerate the loss of atrial kick.
Inotropy may be required for those in frank cardiogenic shock and a failed LV: dobutamine, noradrenaline, adrenaline.
Avoid beta-blockers where possible due to their effect on LV function and HR.
Consider mechanical supports for refractory shock (VA-ECMO, IABP) with the caveat that these may not be appropriate with severe AR (consider LAVA-ECMO).
Surgical consultation, valve surgery is avoided if at all possible in the setting of active IE, but may be required for those with unstable haemodynamics or severe valvular pathology.
Other Cardiac Complications
Septic myocardial infarction (often LAD) can present identical to STEMI
AV block or bradyarrythmia - new rhythm disturbance should raise suspicion for aortic root abscess as the aortic annulus sits next to the AV node.
Septic emboli
have a low threshold to go looking for septic emboli in these patients - if present empiric antibiotic therapy is necessary.
Consider septic CVA in those with headache/neurology. CT angiography and early stroke team input are important. These are at high risk of haemorrhagic transformation.
Consider septic pulmonary emboli in those with right-sided IE
Image for splenic or renal emboli in those with non-specific abdominal pain. Mesenteric or acute limb ischaemia is also possible, as is acute glomerulonephritis.
Thorough assessment of the aortic root and ascending aorta with TOE ± CT angiography if concern for aortitis or aortic root abscess.
Consider toxic shock syndrome which can occur with disseminated Staphylococcal infection (if present consider adding clindamycin, steroids, and IVIG)
DIC is also common with staphylococcal sepsis - screen for it and manage along usual lines.
Get early specialist advice on anticoagulation due to the high risk of bleeding with septic emboli due to the changes in vascular endothelium.